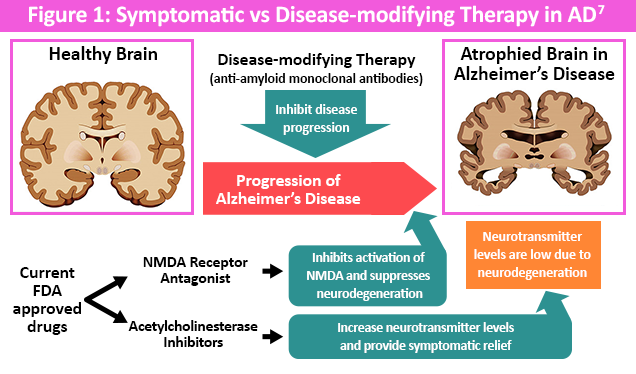
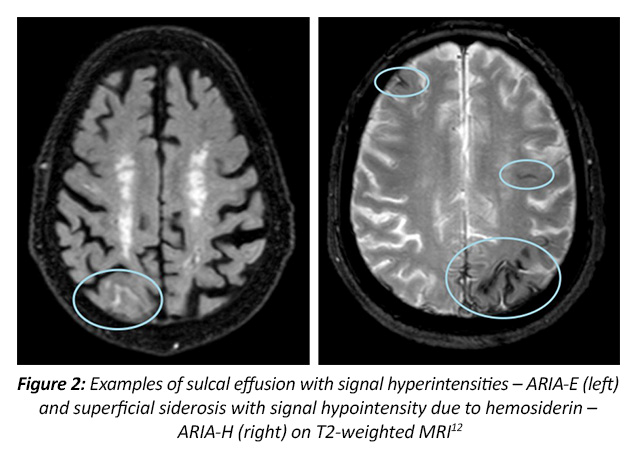
Scientific Council

Marc Agronin, MD
Chief Medical Officer
Frank C. and Lynn Scaduto MIND Institute and Behavioral Health
Miami Jewish Health
Miami, FL

Liana G. Apostolova, MD, MSc, FAAN
Associate Dean of Alzheimer’s Disease Research
Indiana University Distinguished Professor
Barbara and Peer Baekgaard Professor of Alzheimer's Disease Research
Professor in Neurology, Radiology, Medical and Molecular Genetics
Indiana University School of Medicine
Department of Neurology
Indianapolis, IN

Alireza Atri, MD, PhD
Chief Medical Officer, Banner Research
Banner Alzheimer’s and Research Institutes
Pheonix, Sun City, and Tucson, AZ
Director, Banner Sun Health Research Institute
Sun City, AZ

Frederik Barkhof, MD, PhD, FRCR
Professor of Neuroradiology Department of Radiology and Nuclear Medicine Amsterdam UMC (location VU mc) Amsterdam, The Netherlands UCL Queen Square Institute of Neurology UCL Hawkes Institute London, United Kingdom

Sharon A. Brangman, MD, FACP, AGSF
Distinguished Service Professor Chair, Department of Geriatrics Director, Upstate Center of Excellence for Alzheimer’s Disease SUNY Upstate Medical University Syracuse, New York

Christopher Chen, BM BCh, MRCP, FAMS, FRCP
Professor, Department of Pharmacology Director, Memory Aging and Cognition Centre Yong Loo Lin School of Medicine National University of Singapore Singapore

Choong Chih Ching, MBChb NZ, FRCR UK
Senior Consultant, Department of Diagnostic Imaging
RadLink, Singapore
Visiting Consultant, National University Hospital and
National Cancer Centre of Singapore

Anupama Roy Chowdhury, MBBS, MRCP, FAMS
Head and Senior Consultant Department of Geriatric Medicine Singapore General Hospital Singapore

Natalie Christian, MD
Assistant Professor, Geriatrics
Program Director, Geriatric Medicine Fellowship
Tulane University School of Medicine
Southeast Louisiana Veterans Healthcare System
New Orleans, LA

Petrice M. Cogswell, MD, PhD
Associate Professor Mayo Clinic Rochester, Minnesota

Ariel F. Cole, MD, FAAFP
Program Director, AdventHealth Geriatric Fellowship
Winter Park, FL

Brad Dickerson, MD, MMSc, FAAN, FANPA
Professor of Neurology, Harvard Medical School
Director, Frontotemporal Disorders Unit, Department of Neurology, Massachusetts General Hospital
Boston, MA

Richard M. Dupee, MD, MACP, AGSF
Clinical Professor of Medicine
Tufts University School of Medicine
Clinical professor, Department of Public Health and Community Medicine, Tufts University
Chief, Geriatrics Service, Tufts Medical Center
Senior Physician, Pratt Diagnostic Center
Dean ex officio, Office of International Affairs, Tufts University School of Medicine
Boston, MA

Fanny Elahi, MD, PhD
Associate Professor of Neurology, Neuroscience, and AI and Human Health Icahn School of Medicine Mount Sinai New York, NY

James M. Ellison, MD, MPH
Professor of Psychiatry and Human Behavior
Sidney Kimmel Medical College
Co-Director, Comprehensive Alzheimer’s Center
Vickie & Jack Farber Institute for Neuroscience
Thomas Jefferson University
Philadelphia, PA

Juan Fortea, MD, PhD
Professor of Neurology Director, Alzheimer-Down Unit Hospital de la Santa Creu i Sant Pau Sant Pau Biomedical Research Institute Barcelona, Spain

Ana M. Franceschi, MD, PhD
Associate Professor of Radiology Director of Dementia Imaging Director of Molecular Neuroimaging Donald and Barbara Zucker School of Medicine at Hofstra/Northwell Lenox Hill Hospital New York, New York

Giovanni B. Frisoni, MD
Professor of Clinical Neuroscience
University of Geneva
Director, Memory Clinic
Geneva University Hospitals
Geneva, Switzerland

Samuel Gandy, MD. PhD
Professor of Neurology & Psychiatry Icahn School of Medicine at Mount Sinai James J Peters VA Medical Center New York, New York

James E. Galvin, MD, MPH
Professor of Neurology
University of Miami Miller School of Medicine
Miami, FL

David S. Geldmacher, MD
Professor and Director
Division of Memory Disorders and Behavioral Neurology
Department of Neurology
Heersink School of Medicine
University of Alabama at Birmingham
Birmingham, AL

George T. Grossberg, MD
Henry & Amelia Nasrallah Endowed Professor
Director of Geriatric Psychiatry
Department of Psychiatry & Behavioral Neuroscience
Saint Louis University School of Medicine
St. Louis, MO

M. Atsushi Iwata, MD, PhD
Professor of Neurology
Department of Neurology
The University of Tokyo
Tokyo, Japan

Scott A. Kaiser, MD
Director of Geriatric Cognitive Health
Pacific Neuroscience Institute
Santa Monica, CA
Adjunct Professor
USC Leonard Davis School of Gerontology
Los Angeles, CA

Lynn E. Kassel, PharmD, BCPS
Associate Professor of Pharmacy Practice
Drake University College of Pharmacy & Health Sciences
Des Moines, IA

Diana R. Kerwin, MD, CPI
President, Kerwin Medical Center
Chief, Geriatric Medicine, Texas Health Presbyterian Hospital
Dallas, TX

Alka Khera, MD
Assistant Professor
UT Southwestern Neurology
Dallas, TX

Sumeet Kumar, MBBS, DNB, FRCR, EDiNR
Senior Consultant, Department of Neuroradiology National Neuroscience Institute of Singapore Clinical Assistant Professor, Duke-NUS Medical School Adjunct Director for Advanced Magnetic Resonance Imaging Research Radiological Sciences Academic Clinical Program (RADSC ACP) SingHealth Duke-NUS Academic Medical Centre Singapore

Rosemary D. Laird, MD, MHSA, AGSF
Chief Medical Officer, My Memory Clinic
Principal Investigator, ClinCloud Clinical Trials
Viera, Florida

Chi-Ying (Roy) Lin, MD, MPH, FAAN
Director, CurePSP Center of Care for PSP, CBD, and MSA
Assistant Professor of Neurology
Alzheimer's and Parkinson's Disease Centers
Baylor College of Medicine
Houston, TX

Catherine A. Madison, MD
Founding Director Ray Dolby Brain Health Center
San Francisco, CA

Scott McGinnis, MD
Assistant Professor of Neurology, Harvard Medical School
Center for Alzheimer Research and Treatment
Brigham and Women's Hospital
Frontotemporal Disorders Unit
Massachusetts General Hospital
Boston, MA

Kristin S. Meyer, PharmD, BCGP, FASCP
Professor of Pharmacy Practice
Drake University College of Pharmacy and Health Sciences
Des Moines, IA

Charles Montano, MD
Owner, Principal Investigator
CT Clinical Research
Cromwell, CT

Mary Norman, MD
Geriatrician
Cedars-Sinai Medical Group
Los Angeles, CA

Tiago Gil Oliveira, MD, PhD
Associate Professor Life and Health Sciences Research Institute School of Medicine University of Minho Neuroradiologist Hospital de Braga Braga, Portugal

Monica Parker, MD
Associate Professor of Neurology
Lead, Outreach, Recruitment, and Engagement Core
Goizueta Alzheimer's Disease Research Center
Goizueta Brain Health Institute
Emory University
Atlanta, Georgia

Lucilla Parnetti, MD
Professor of Neurology Department of Medicine and Surgery University of Perugia Perugia, Italy

Manisha Parulekar, MD, FACP, AGSF, CMD
Director, Division of Geriatrics
Co-director, Center for Memory Loss and Brain Health
Hackensack University Medical Center
Hackensack, NJ

Giulio Maria Pasinetti, MD, PhD
The Saunders Family Chair in Neurology, Professor in Neurology
Director, Mount Sinai Center in Advance Botanical Research in Molecular Integrative Neuroresilience
Icahn School of Medicine at Mount Sinai
New York, NY
Director, Basic and Biomedical Research and Training Program, Geriatric Research and Clinical Center
James J. Peters Department of Veterans Affairs Medical Center
Bronx, NY

Robert Perneczky, MD, PhD
Professor of Psychiatry Department of Psychiatry and Psychotherapy Ludwig-Maximilians-University Munich German Center for Neurodegenerative Diseases Munich, Germany

Anton P. Porsteinsson, MD
William B. and Sheila Konar Professor of Psychiatry, Neurology,
Neuroscience, and Medicine
Director, Alzheimer’s Disease Care, Research and Education
Program (AD CARE)
University of Rochester School of Medicine and Dentistry
Rochester, NY

Michael S. Rafii, MD, PhD
Professor of Clinical Neurology Medical Director, Alzheimer’s Therapeutic Research Institute Keck School of Medicine of USC Los Angeles, California, USA

William D. Rhoades, DO, FACP
Chief Medical Officer
Advocate Good Samaritan Hospital
Downers Grove, IL

Barry W. Rovner, MD
Professor, Departments of Psychiatry & Neurology
Sidney Kimmel Medical College
Thomas Jefferson University
Philadelphia, PA

Marwan Noel Sabbagh, MD, FAAN, FANA
Moreno Family Chair for Alzheimer’s Research
Vice Chairman for Research and Professor
Department of Neurology
Barrow Neurological Institute
Phoenix, AZ

Martin J. Sadowski, MD, PhD
Zachary and Elizabeth M. Fisher Professor of Neurodegeneration and Alzheimer's Disease Professor of Neurology, Psychiatry, and Biochemistry & Molecular Pharmacology New York University Grossman School of Medicine Director, Fisher Center Alzheimer’s Clinical Trials Program New York, NY

Stephen Salloway, MD, MS
Professor, Psychiatry and Human Behavior Professor of Neurology, Alpert Medical School of Brown University Providence, Rhode Island

Angela Sanford, MD, CMD
Associate Professor of Internal Medicine-Geriatrics
Saint Louis University School of Medicine
Ballwin, MO

Philip Scheltens, MD, PhD
Professor Emeritus of Neurology Amsterdam University Medical Center Head Dementia Fund at EQT Life Sciences Amsterdam, Netherlands

Suzanne E. Schindler, MD, PhD
Associate Professor of Neurology Division of Neurology Washington University School of Medicine in St. Louis Investigator, Knight Alzheimer Disease Research Center St. Louis, Missouri, USA

Paul E. Schulz, MD
Rick McCord Professor of Neurology
Umphrey Family Professor of Neurodegenerative Disorders
Director, Memory Disorders and Dementia Clinic
UTHealth Houston
Houston, TX

Sharon J. Sha, MD, MS
Clinical Professor Clinical Professor Neurology and Neurological Sciences Stanford University Chief, Stanford Memory Disorders Center Stanford Medicine Health Care Palo Alto, CA

Jiong Shi, MD
Professor of Neurology
Department of Neurology
Huashan Hospital
Fudan University
Shanghai, China

Neil Skolnik, MD
Professor of Family and Community Medicine
Sidney Kimmel Medical College
Thomas Jefferson University
Philadelphia, PA

Sandra Swantek, MD
Director, Section of Geriatric Psychiatry
RUSH University Medical Center
Chicago, IL

R. Scott Turner, PhD, MD
Professor of Neurology
Director of the Memory Disorders Program
Georgetown University
Washington, DC

Lawren VandeVrede, MD, PhD
Assistant Professor of Neurology Memory and Aging Center Department of Neurology University of California, San Francisco San Francisco, California, USA

Chuck Vega, MD, FAAFP
Health Sciences Clinical Professor
UC Irvine Department of Family Medicine
Director, UCI Program in Medical Education for the Latino Community
University of California Irvine
Irvine, CA

Nicolas Villain, MD, PhD
Professor of Neurology Department of Neurology Sorbonne University Neurologist, Pitié-Salpêtrière Hospital Assistance Publique – Hôpitaux de Paris Paris, France

Geoffrey C. Wall, PharmD, FCCP, BCPS
John R. Ellis Distinguished Chair of Pharmacy Practice
Professor of Clinical Sciences
Director, Drake Drug Information Center
Drake University College of Pharmacy and Health Sciences
Internal Medicine Clinical Pharmacist
Iowa Methodist Medical Center
Des Moines, IA

David A. Wolk, MD, FAAN
Professor of Neurology
Director, Penn Alzheimer’s Disease Research Center
University of Pennsylvania
Philadelphia, PA