Blood‐Based Biomarker Tests
Adoption of a biomarker-enabled care pathway for Alzheimer’s disease in primary care
Blood-based biomarkers for Alzheimer’s disease (AD) are increasingly recognized as an important tool in the evaluation of patients presenting with cognitive symptoms. When used appropriately, biomarkers can clarify underlying AD pathology, reduce diagnostic uncertainty, and support timely referral, prognosis, and follow-up. To address the growing need for practical, clinician-focused guidance, a multidisciplinary AD Biomarker Task Force was convened. 1 The Task Force recognized that primary care practitioners (PCPs) play a critical role in the identification, diagnosis, and ongoing management of patients with AD, and that educational approaches to biomarker use must be tailored to the realities of primary care practice (Figure 1).1
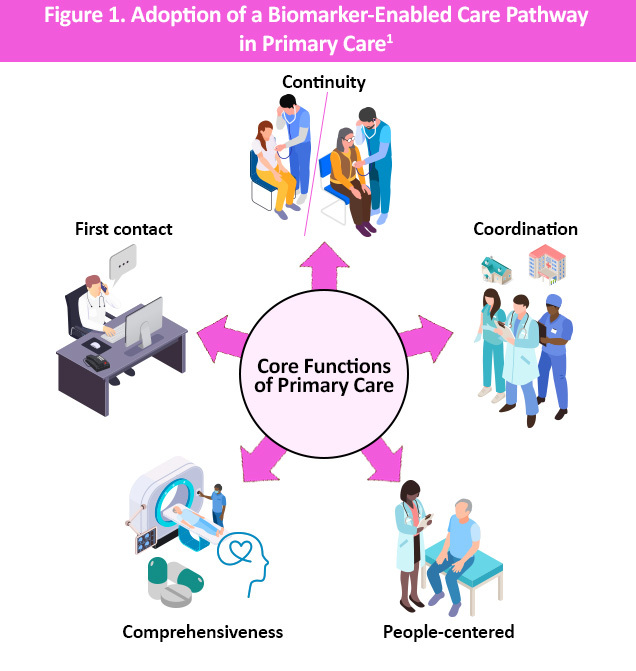
The Task Force concluded that adoption of a biomarker-enabled care pathway in primary care is essential to improving early detection of AD, supporting appropriate referral to specialists, and enabling coordinated, longitudinal care.
Overview of blood-based biomarkers
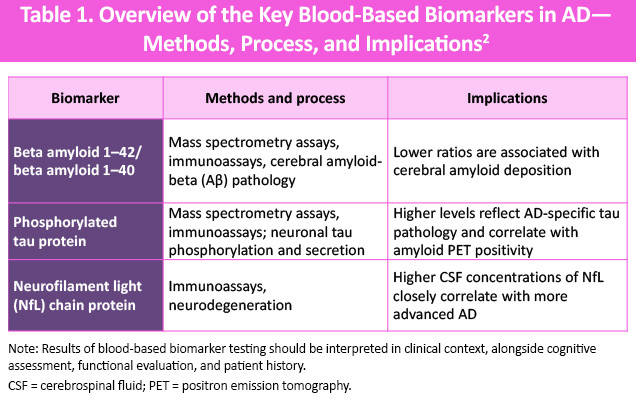
Blood-based biomarkers offer a scalable and minimally invasive approach to detecting AD-related pathology and may improve access to biologic testing, particularly in settings where advanced imaging or cerebrospinal fluid (CSF) testing is limited. The key biomarkers that have been identified include amyloid, phosphorylated tau protein, and neurofilament light (NfL) protein (Table 1).2
How biomarkers fit into AD care models
A significant advancement in AD diagnostics in recent years has been the development of methods to detect and measure blood-based markers, with some assays exhibiting accurate diagnostic performance. This approach makes the biological diagnosis of AD more accessible compared with PET or CSF assays. Therefore, the use of blood-based markers is projected to revolutionize clinical care and research. 3
Core biomarker framework
To support practical clinical application, biomarkers are grouped into 2 categories (Table 2)
- Core 1 biomarkers: Amyloid (A), early tau (T1), and hybrid combinations
- Core 2 biomarkers: Tau PET imaging and later-changing tau (T2) biofluid biomarkers
The diagnosis of AD can be established by abnormality in specific Core 1 biomarkers, which include amyloid PET, CSF Aβ42/40, CSF p-tau181/Aβ42, and CSF total tau/Aβ42, and can be validated via accurate plasma-based assays. On May 16, 2025, the US Food and Drug Administration (FDA) granted its first clearance to a blood test that utilizes the plasma p-tau217/Aβ1–42 ratio to diagnose AD in symptomatic patients aged 55 years or older.4 Thus, the recommendation is that the different Core 1 biomarkers should be interchangeable for the detection of AD neuropathologic change (ADNPC) and consequently for diagnosing AD. As nearly all symptomatic individuals and the vast majority of asymptomatic individuals with abnormal amyloid PET will have intermediate/high ADNPC, the Core 1 category represents ADNPC more generally (ie, both plaques and tangles). 3

Biomarker-enabled AD diagnostic care pathway
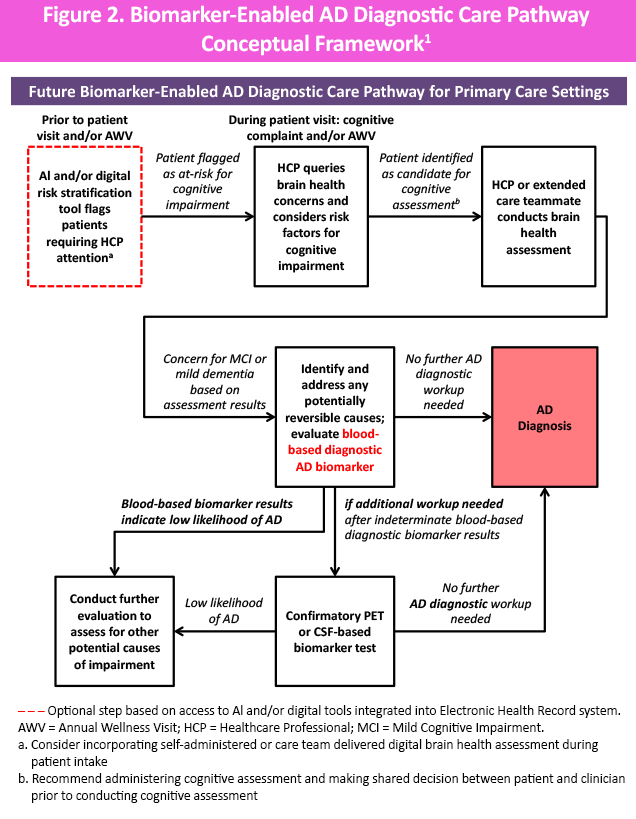
Biomarkers are most effective when integrated into a structured diagnostic care pathway that begins in primary care and incorporates specialty collaboration when needed. The recommended pathway emphasizes early identification of cognitive concern, strategic use of blood-based biomarkers, targeted use of imaging or CSF testing, timely referral to neurology or memory care specialists, and ongoing monitoring and follow-up. The Task Force’s proposed diagnostic care pathway envisions using artificial intelligence (AI) risk stratification before a visit to identify patients at higher risk of AD or dementia, followed by triage to appropriate providers for digital or clinician-led brain health assessment during the visit (Figure 2).1
Multidisciplinary collaboration
Optimal AD care depends on clear coordination between PCPs, neurologists, geriatricians, neuroradiologists, pathologists, psychiatrists, and other stakeholders. While PCPs are central to early identification and longitudinal management, consensus is needed across disciplines to determine optimal strategies for systematizing biomarker use, integrate tools into existing clinical decision support infrastructure, and reduce barriers to adoption of biomarker-enabled care models. Rapid advances in the AD care landscape—including the availability of disease-modifying therapies (DMTs), blood-based biomarkers for AD, and the launch of the Centers for Medicare & Medicaid Services (CMS) Innovation Center Guiding an Improved Dementia Experience (GUIDE) Model—underscore the importance of expert consensus on the role of blood-based biomarkers in clinical workflows.1

Summary
Drawing on findings from published literature and clinical expertise, the AD Biomarker Task Force identified opportunities to promote systematic utilization of blood-based biomarkers and the adoption of a biomarker-enabled diagnostic care pathway in primary care. These approaches are intended to support earlier identification of patients with AD, facilitate appropriate referral, and improve coordination across care teams. Effective use of biomarkers has the potential to improve care delivery and reduce inequities driven by limited access to diagnostic testing.
References
- Borson S, Au R, Chodos AH, et al. Opportunities to encourage adoption of a biomarker-enabled care pathway for Alzheimer’s in primary care. Alzheimers Dement (Amst). 2025;17:e70095.
- Arslan B, Zetterberg H, Ashton NJ. Blood-based biomarkers in Alzheimer’s disease – moving towards a new era of diagnostics. Clin Chem Lab Med. 2024;62:1063-1069.
- Jack CR Jr, Andrews SJ, Beach TG, et al. Revised criteria for the diagnosis and staging of Alzheimer’s disease. Nat Med. 2024;30:2121-2124.
- Hu S, Yu H, Gao J. The pTau217/Aβ1-42plasma ratio: the first FDA-cleared blood biomarker test for diagnosis of Alzheimer’s disease. Drug Discov Ther. 2025;19(3):208-209.