ARIA
Amyloid-related imaging abnormalities (ARIA) are radiographic findings resembling cerebral amyloid angiopathy–related inflammation and have been shown to occur in approximately 10% to 30% of all patients receiving amyloid immunotherapy for the treatment of Alzheimer’s disease (AD). ARIA can present as ARIA-E (edema/effusion) or ARIA-H (microhemorrhages/superficial siderosis) and may be asymptomatic or, in rare cases, symptomatic, involving neurologic symptoms such as headache, confusion, dizziness, visual changes, or praxis difficulties. In these cases, a critical concern is differentiating symptomatic ARIA from stroke, as misdiagnosis and treatment with thrombolytics can potentially result in fatal intracranial hemorrhage.1
Pathophysiology & Classification
The proposed mechanism is that ARIA occurs due to the removal of amyloid deposits from and alongside cerebral arteries, resulting in vessel leakage and potentially triggering an immune response that can continue for weeks to months, even after cessation of anti-amyloid immunotherapy (Figure 1).1
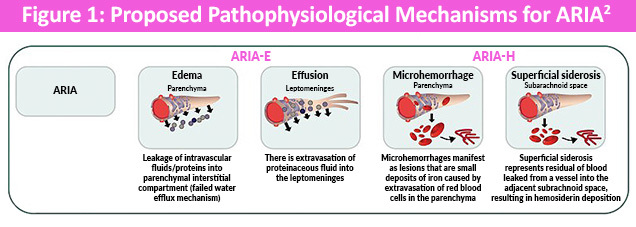
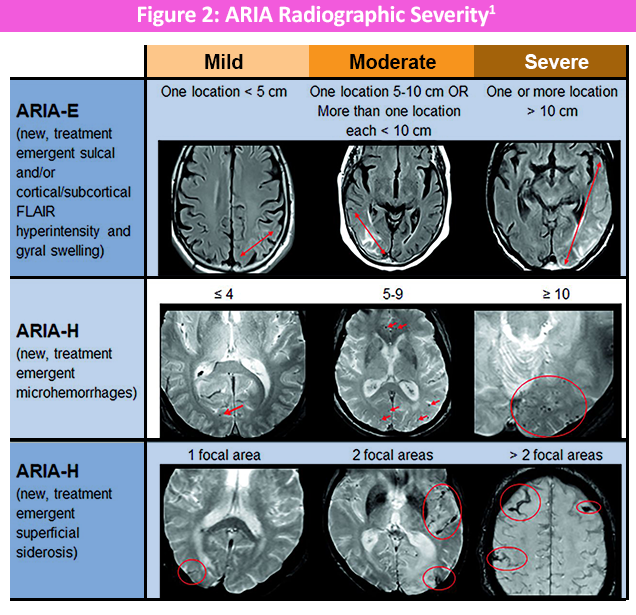
In terms of the classification, the 2 types have been named ARIA-E and ARIA-H. ARIA-E, in which “E” stands for vasogenic edema and sulcal effusions, is characterized as the extravasation of fluid resulting in interstitial vasogenic edema or sulcal effusion in the leptomeningeal subpial space. ARIA-H, in which “H” stands for hemorrhage, includes microhemorrhages and superficial siderosis that, in rare cases, may be associated with intraparenchymal hemorrhage and intraventricular hemorrhage. The US Food and Drug Administration (FDA) has adopted a standard radiographic classification for ARIA, which is divided into 3 severity categories (mild, moderate, and severe), based on the size and site of involvement in ARIA-E and the number of microhemorrhages or focal areas involved in ARIA-H as shown in Figure 2.1,2
Clinical Presentation & Imaging
Data from clinical trials indicate that 74% to 97% of ARIA are asymptomatic. When ARIA is symptomatic, clinical manifestations typically present as headache or confusion. Less frequently, patients may experience visual disturbances, visuospatial impairment, or praxis difficulties, reflecting the relative predilection of ARIA-E for posterior brain regions. Consistent with imaging findings, clinical symptoms generally resolve over time, particularly with treatment pause or discontinuation. In the uncommon event of symptomatic ARIA-E, or in cases of asymptomatic but radiographically severe ARIA-E, appropriate clinical management should be initiated and continued until symptom resolution.1,3
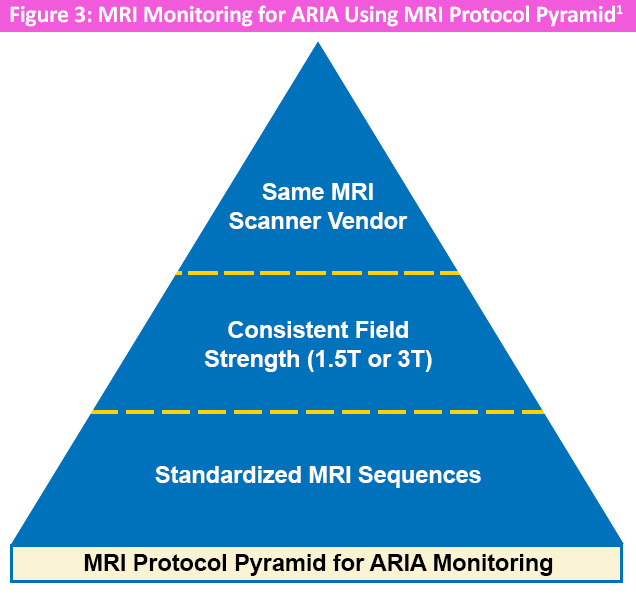
Standardized magnetic resonance imaging (MRI) protocols are essential for ARIA evaluation because they enable the application of consistent assessment and treatment criteria and facilitate reliable comparison across serial MRI examinations within an individual patient. Ideally, patients should be imaged using the same magnetic field strength, scanner vendor, and scanner model, with identical sequences and sequence parameters across follow-up studies, as illustrated in Figure 3.
Monitoring Guidelines
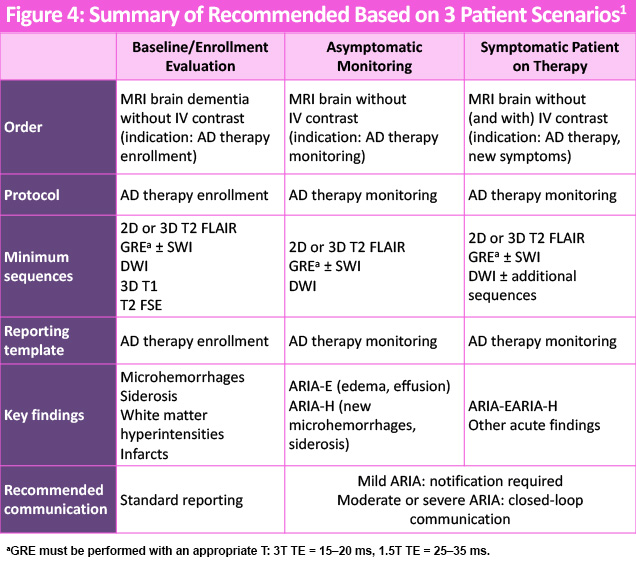
MRI protocols per the current recommendations vary depending on the presence or absence of symptoms. Accordingly, distinct imaging orders may be considered for baseline/enrollment assessment, routine monitoring of asymptomatic ARIA, and evaluation of symptomatic patients receiving anti-amyloid immunotherapy. These scenarios can be aligned with specific imaging protocols and standardized reporting templates, as outlined in Figure 4.
Case Escalation Flowchart
Currently, 2 anti-amyloid agents are approved for the treatment of Alzheimer’s disease (AD): donanemab and lecanemab. To support safe and consistent clinical use, a multidisciplinary expert committee was convened to develop recommendations for ARIA monitoring schedules, escalation pathways, and management strategies for patients receiving these therapies.
Click here to access the case escalation flowchart for these agents.
Shared Decision-Making (SDM) Guide: Discussing ARIA With Patients & Caregivers
Click here to access and download an easy-to-use shared decision-making guide designed to support clear, patient-centered discussions about ARIA with patients and caregivers.
References
- Cogswell PM, Andrews TJ, Barakos JA, et al. Alzheimer disease anti-amyloid immunotherapies: imaging recommendations and practice considerations for monitoring of amyloid-related imaging abnormalities. AJNR Am J Neuroradiol. 2025;46:24-32.
- Hampel H, Elhage A, Cho M, et al. Amyloid-related imaging abnormalities (ARIA): radiological, biological and clinical characteristics. Brain. 2023;146:4414-4424.
- Cogswell PM, Barakos JA, Barkhof F, et al. Amyloid-related imaging abnormalities with emerging Alzheimer disease therapeutics: detection and reporting recommendations for clinical practice. AJNR Am J Neuroradiol. 2022;43:E19-E35.